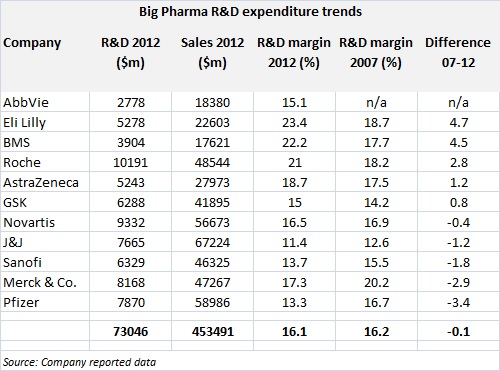
And here's a real kicker:
http://www.businessweek.com/article...ug-delays-push-novartiss-diovan-prices-higher
Basically US patent law allows for a single company to win monopoly to manufacture the generic drug for the first six months. And here is the biggest outrage. If that company can't or won't manufacture it safely, everyone else is blocked, by US law, from manufacturing it, even if they could do so safely, and it effectively remains a monopoly for the original patent holder, indefinitely. This is a rigged system to fleece Americans.
Didn't know that. Seems pretty screwed up. I could understand permitting an exclusivity arrangement with a single company to mitigate the loss of profit the "real drug" company would have after the drug can go generic, but I think it is pretty lame that they can avoid producing the drug during the exclusivity period they are granted. I think they should have a mandatory quota to be met, or something similar.